Tag: Sentinel System

How to Track Post-Marketing Studies for Drug Safety
Learn how post-marketing studies track drug safety after approval, using FAERS, Sentinel, and real-world data to catch hidden risks. Understand the systems, challenges, and future of pharmacovigilance.
view morerecent posts
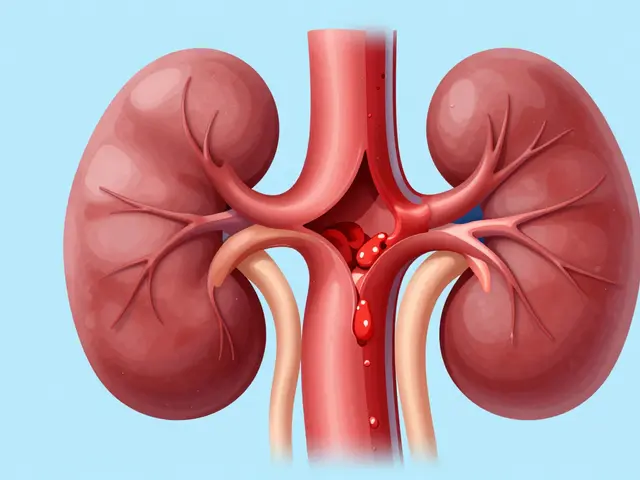
Why ACE Inhibitors Are Risky for Renal Artery Stenosis
Learn why ACE inhibitors are contraindicated for bilateral renal artery stenosis and how these drugs affect kidney filtration and blood pressure.
Published ON: 12 Apr
Cognitive Biases: How Beliefs Shape Your Decisions and Responses
Explore how cognitive biases and deep-seated beliefs systematically alter your decision-making and generic responses, backed by neuroscience and real-world data.
Published ON: 1 Apr
Managing Opioid-Induced Constipation: Prevention and Treatment Guide
Learn how to prevent and treat Opioid-Induced Constipation (OIC). Explore the difference between standard laxatives and PAMORAs for lasting relief.
Published ON: 8 Apr